A practical guide for procurement leaders managing compliance-critical translation projects
Why Translation Accuracy Is Critical for Regulatory Compliance in Multinational Organisations
Picture this: Your organisation is six weeks from launching a regulated product across five European markets. The documentation stack spans legal contracts, regulatory submissions, product labelling, patient information leaflets, and internal SOPs. Legal, compliance, regulatory affairs, R&D, and marketing are all converging on immovable deadlines — and procurement has just been asked to sign off on the translation partner.
This is the moment when theoretical risk becomes tangible.
A mistranslation in a pharmaceutical patient leaflet isn’t an embarrassment — it’s a regulatory violation that can halt distribution. An error in a financial disclosure doesn’t just damage reputation — it triggers compliance investigations. In legal contracts, ambiguity doesn’t get forgiven; it gets litigated. Yet the procurement landscape offers no shortage of Language Service Providers (LSPs) claiming quality, compliance expertise, and speed. The real question isn’t who says they can deliver — it’s how you verify their quality control mechanisms actually satisfy regulatory standards.
This distinction has never mattered more. As global expansion accelerates, regulatory frameworks tighten, and AI enters translation workflows at scale, procurement leaders face a paradox: drive efficiency without compromising the one thing regulators won’t negotiate on — accuracy. This article maps out how leading procurement teams evaluate translation quality in regulated environments, focusing on the control mechanisms that withstand both operational pressure and regulatory scrutiny.
Why Translation Quality Control Is Now a Strategic Priority for Procurement Teams in Regulated Industries
Translation used to be treated as a commodity purchase. Select a vendor, provide source content, receive target content. That model has collapsed under the weight of regulatory complexity.
The Three Key Forces Changing How Procurement Evaluates Translation Providers
1. How Overlapping Global Regulations Increase Translation and Compliance Risk
Organisations now navigate overlapping jurisdictions: EU MDR for medical devices, FDA requirements for pharmaceuticals, GDPR for data privacy, FCA rules for financial services. Documentation must be accurate not just at publication, but traceable and defensible years later when regulators ask questions.
2. Why Speed-to-Market Pressures Make Translation Quality Control More Critical Than Ever
Regulatory timelines may be long, but competitive windows aren’t. Procurement teams are expected to enable rapid global launches while maintaining absolute accuracy — a tension that breaks poorly designed quality systems.
3. How Poorly Governed AI Translation Creates Hidden Compliance Risks
Machine translation is now embedded in most enterprise translation ecosystems. When governed well, it accelerates delivery without sacrificing quality. When governed poorly, it introduces systemic risk that only surfaces during audits or post-market surveillance.
Against this backdrop, quality control has shifted from operational detail to strategic procurement criterion. The challenge is making it measurable.
Why Procurement Teams Struggle to Evaluate Translation Quality in Regulated Environments
Most RFPs for translation services include a standard question: “Please describe your quality assurance process.” The responses are predictably reassuring. Certified linguists. Multi-stage review. Proven workflows. Yet procurement teams struggle to distinguish between:
- Marketing language and operational reality
- Individual translator competence and systemic quality control
- Compliance-ready processes and best-effort approaches
The Translation Quality Gaps That Create Regulatory and Commercial Risk
How Inconsistent Translation Quality Across Languages Increases Regulatory Exposure
An LSP may excel in French and German while struggling with Polish or Finnish — exactly where regulatory exposure can be highest for markets with limited vendor depth.
Why Lack of Workflow Transparency Fails Regulatory Audits
Without visibility into who reviewed what, when, and how disputes were resolved, procurement is left trusting assurances rather than evidence. Regulators don’t accept assurances.
How Low-Cost Translation Procurement Can Increase Compliance Risk
Lower per-word pricing looks attractive in a tender. But rework, delayed approvals, or compliance failures erase savings quickly — and create costs procurement doesn’t typically track.
Why Procurement Must Align Legal, Compliance and Marketing Translation Requirements
Legal needs traceability. Compliance needs auditability. Marketing needs brand consistency. Procurement must reconcile all three into a single vendor decision, often without a shared framework.
This is where understanding what quality control actually means in a regulatory context becomes essential.
What Translation Quality Control Really Means for Regulatory Compliance
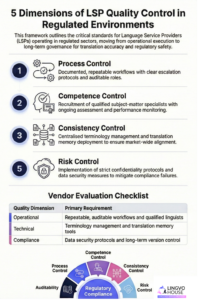
The five dimensions procurement teams should assess when evaluating LSP quality control mechanisms for compliance-critical translation projects. Download the full framework guide.
The Five Core Components of Translation Quality Control in Regulated Industries
1. Why Documented Translation Processes Are Essential for Regulatory Compliance
Documented, repeatable workflows that define:
- Who translates and based on what qualification criteria
- Who reviews and what their mandate includes
- Who approves final output and under what conditions
- How issues escalate when reviewers disagree
2. How Linguist Qualifications and Subject-Matter Expertise Affect Compliance Outcomes
Mechanisms ensuring linguists are:
- Professionally qualified (not just native speakers)
- Subject-matter specialists in the relevant domain
- Assessed at onboarding and monitored over time
3. Why Terminology Consistency Is Critical for Regulatory Submissions
Systematic use of terminology management, translation memory, and style guides to maintain consistency across markets, document types, and time — critical when regulatory bodies cross-reference submissions.
4. How Translation Risk Management Protects Confidentiality and Regulatory Compliance
Proactive identification and mitigation of risks related to confidentiality, data security, and regulatory non-compliance — including contingency protocols when deadlines compress.
5. Why Audit Trails and Traceability Matter in Regulated Translation Projects
The ability to demonstrate, years after delivery, that appropriate controls were applied. This means version control, decision logs, and clear accountability trails.
When procurement evaluates LSPs through this lens, quality becomes verifiable rather than subjective.
A Practical Procurement Framework for Evaluating Translation Quality Control
Below is a procurement-friendly framework for assessing whether an LSP’s quality mechanisms satisfy regulatory requirements.
1. How to Evaluate ISO 17100 and ISO 9001 in Real Translation Workflows
Certifications like ISO 17100 (translation services) and ISO 9001 (quality management) are valuable baseline indicators. They confirm documented processes, qualified personnel, and continuous improvement commitments.
But procurement should ask:
- How are these standards embedded in day-to-day delivery, not just policy documents?
- Are they applied across all services or limited to certain tiers?
- What happens when deadlines compress—do quality controls flex or hold?
Standards prove systems exist. Application proves they function under pressure.
2. How Four-Eyes Review Models Protect Translation Quality in Regulated Content
Key questions for translation quality assurance:
- Is revision by a second qualified linguist mandatory, or conditional?
- Are reviewers selected based on subject-matter expertise, not just language availability?
- How are discrepancies between translator and reviewer resolved and documented?
In regulated work, “four-eyes review” isn’t optional — it’s foundational. But not all four-eyes models are equal. The difference lies in reviewer selection criteria and dispute resolution protocols.
3. How Terminology Management Reduces Regulatory Risk in Multilingual Documentation
Regulatory terminology isn’t static. Classifications change. Approved terms evolve. Inconsistent terminology creates compliance exposure.
Ask how the LSP:
- Builds and maintains client-specific, domain-specific glossaries
- Manages terminology updates when regulatory guidance shifts
- Ensures consistent usage across markets and over time
- Prevents terminology drift when multiple linguists work on related projects
Strong terminology governance doesn’t just improve quality — it reduces the regulatory surface area for error.
4.How to Assess AI Translation Governance in Regulated Environments
AI-assisted translation can improve efficiency when governed rigorously. But in regulated environments, “efficiency” without control creates liability.
Procurement should clarify:
- Where machine translation is used and where it’s prohibited
- When human post-editing is mandatory versus optional
- How quality thresholds are defined and monitored for AI-assisted content
- Whether clients can configure AI usage by content type or risk profile
The most mature LSPs treat AI as a tool within a governed quality framework—not a cost reduction shortcut that shifts risk to the client.
5. Why Real-Time Translation Tracking and Audit Trails Matter for Compliance
Modern translation quality control isn’t a black box. Leading providers offer:
- Real-time project tracking with stage-by-stage visibility
- Clear audit trails showing who touched what and when
- Access to historical versions and decision logs
This transparency supports both compliance teams managing current projects and audit teams investigating past ones. It also shifts accountability from trust to evidence.
How Translation Quality Control Will Shape Procurement Strategy Over the Next Five Years
Procurement leaders selecting LSPs today are making decisions that will shape compliance infrastructure for years. Three trends are worth anticipating:
1. Why Data-Driven Translation Quality Metrics Will Become a Procurement Standard
Expect greater adoption of:
- Linguistic KPIs tracked across projects and languages
- Quality scoring models that aggregate reviewer feedback, client corrections, and delivery metrics
- Continuous performance benchmarking against SLAs
This shift moves procurement from anecdotal feedback (“the team was happy”) to measurable outcomes that satisfy both finance and compliance stakeholders.
2. How Technology-Enabled Translation Compliance Will Define Future LSP Selection
Regulators are increasingly comfortable with technology—provided governance is demonstrable. LSPs that can prove technology-enabled compliance (not just technology-enabled speed) will gain competitive advantage.
This includes showing how AI improves consistency without reducing human oversight, and how automation enhances traceability rather than obscuring it.
3. Why Configurable Translation Workflows Will Replace One-Size-Fits-All Models
One-size-fits-all workflows won’t suffice in multi-stakeholder organisations. Future-proof LSPs will offer configurable quality levels based on:
- Content type (e.g., regulatory submissions vs. internal training materials)
- Risk profile (high-risk patient-facing content vs. low-risk operational documents)
- Market maturity (established markets vs. emerging ones)
This configurability allows organisations to optimise cost intelligently — applying maximum controls where risk is highest, without over-engineering low-risk workflows.
How Leading Organisations Manage Translation Quality in Compliance-Critical Sectors
Across pharmaceuticals, legal services, and financial institutions, a clear pattern emerges among organisations that successfully manage multilingual compliance.
They don’t rely on heroic individual effort or reactive quality checks. They partner with LSPs who embed quality control into every stage of the translation lifecycle — combining ISO standards, domain expertise, technology governance, and auditability into a single, coherent system.
From procurement’s perspective, this approach delivers three strategic advantages:
- Simplified vendor management: One partner, one quality framework, less internal coordination overhead
- Reduced downstream risk: Fewer regulatory queries, faster approvals, lower rework costs
- Stakeholder confidence: Legal, compliance, and regulatory teams trust the output, reducing internal friction
It also shifts the conversation from price per word to total cost of ownership—factoring in risk mitigation, speed-to-market, and regulatory confidence.
Why Translation Quality Control Is a Competitive Advantage for Procurement Teams
Selecting an LSP for compliance-critical work is no longer a tactical sourcing exercise. It’s a strategic decision that shapes regulatory outcomes, operational resilience, and brand reputation across markets.
By focusing on quality control mechanisms that satisfy regulatory standards — process control, competence control, consistency control, risk control, and auditability — procurement leaders can:
- Reduce compliance exposure before it reaches auditors
- Build internal stakeholder confidence in global expansion decisions
- Enable faster, safer market entry without regulatory compromise
The challenges are real. But they’re solvable with the right frameworks and the right partners.
If you’d like to explore how these frameworks could support your procurement strategy for regulatory translation, the team at LingvoHouse is available for a conversation. For a practical next step, use our instant quote platform to benchmark workflows, costs, and quality controls for your upcoming projects.
Translation Quality Control for Regulatory Compliance: Frequently Asked Questions
What is ISO 17100 and why does it matter for translation quality?
ISO 17100 is the international standard for translation services that defines requirements for the core translation process, resources, and quality management systems. For regulated industries, ISO 17100 certification demonstrates that an LSP has documented processes, qualified linguists, and mandatory review protocols — essential for compliance and auditability.
How do you evaluate translation quality in pharmaceutical projects?
Pharmaceutical translation quality evaluation should assess:
(1) linguist qualifications in medical/pharma terminology
(2) mandatory four-eyes review by subject-matter experts
(3) terminology management systems for regulatory consistency
(4) audit trails for regulatory submissions
(5) compliance with ISO 17100 and ISO 9001 standards
What’s the difference between machine translation and AI-assisted translation in regulated environments?
Machine translation generates automated translations without human review, which is inappropriate for regulatory content. AI-assisted translation uses machine translation with mandatory human post-editing by qualified linguists — when properly governed, this can improve efficiency while maintaining compliance standards through documented review processes.
How can procurement verify LSP quality claims during vendor selection?
Request evidence of: ISO certifications and their scope, sample audit trails from past projects, linguist qualification processes, terminology management protocols, quality metrics and KPIs, and references from similar regulated clients. Ask how quality controls are maintained under deadline pressure — where generic promises often reveal operational reality.
What are the risks of choosing low-cost translation providers for compliance documents?
Low-cost providers often lack: mandatory review processes, subject-matter specialist linguists, terminology governance systems, and audit trail capabilities. This creates regulatory exposure through inconsistent terminology, mistranslations in critical documents, delayed approvals, potential recalls, and missing documentation during regulatory audits — costs that far exceed initial savings.







